Trending...
- "Stop scrolling and start watching" - Beloved film recommendation site Criticker gets a major makeover
- Green Energy Solar Expands with New Offices in Port St. Lucie, West Palm Beach, and Orlando
- xREnergy up as much as +3,094,634% on first day listed on the XRP Ledger. Ticker : $XRE
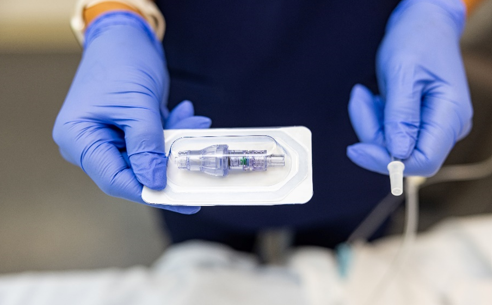
FAYETTEVILLE, Ark. - AussieJournal -- Lineus Medical® is proud to announce the U.S. Food and Drug Administration (FDA) has cleared its flag ship product, SafeBreak® Vascular, for use on all vascular access lines including peripheral IV catheters, midlines, peripherally inserted central catheters, central venous catheters, IV ports, and interosseous vascular access devices on patients two weeks of age and up. This allows SafeBreak Vascular to be installed on both adults and children and on every type of IV line.
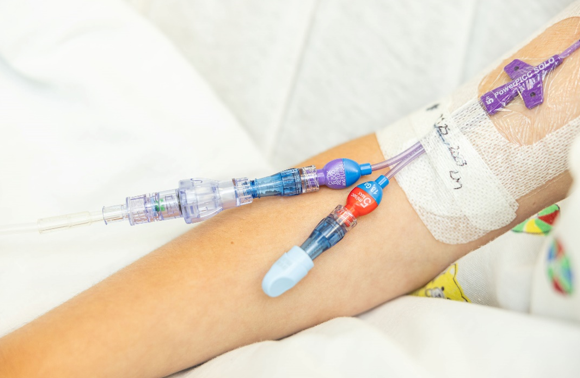
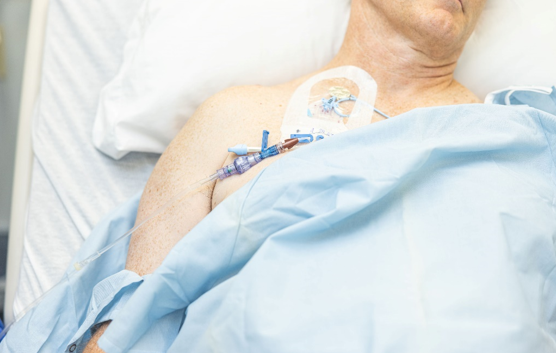
SafeBreak Vascular is the only breakaway device for IV lines proven to reduce IV complications.1 When a harmful force is placed on the line, SafeBreak intentionally separates to remove the damaging force and protect the patient's IV. When SafeBreak separates, valves on both ends of the device close preventing medicine spills from the pump and blood loss from the patient. The separated device also causes the IV pump to alarm, notifying the nursing staff to check on the patient. To replace SafeBreak, each component is unthreaded from the line, a new sterile SafeBreak is installed, and the patient's infusion is restarted. Patients avoid additional needlesticks, nurses save time, and hospitals save money1. It's a win-win-win.
More on Aussie Journal
Ryan Brick, VP of Sales, stated, "This is a large milestone for our company and a steppingstone towards our mission of reducing IV-line failure. With SafeBreak's clearance for all vascular lines, we can help protect every patient user and every IV line throughout the entire country. Sales momentum has continued to build since our pediatric clearance this past May, and now, the expansion of SafeBreak's indications will bring even more interest and help introduce the product to more hospitals. We will continue to work to make SafeBreak the standard of care for every hospital and patient."
Vance Clement, CEO of Lineus Medical, stated, "What an exciting time for SafeBreak Vascular and Lineus Medical! We recently gained licence in Canada and now FDA clearance for all medical lines for patients to two weeks of age and up in the United States. SafeBreak is a simple device that can make a big difference for nurses and hospitals. These new indications are for more complicated and more expensive IV lines that are used for long-term infusions, cancer patients, and in emergency situations. SafeBreak has been shown to work well on peripheral IVs, and now it can be used by nurses to protect these more expensive lines on more vulnerable patient populations. With these additional indications, we can expand the benefit of SafeBreak throughout the entire hospital."
More on Aussie Journal
ABOUT LINEUS MEDICAL
Lineus Medical is the developer of a break-away technology that is proven to reduce IV restarts and IV complications1. Our mission is to remove the pains associated with medical lines. More information about Lineus Medical can be found on the company website: www.lineusmed.com or you can follow the company on Facebook or LinkedIn.
1. Data on File
MKG-0237 Rev 00
SafeBreak Vascular is the only breakaway device for IV lines proven to reduce IV complications.1 When a harmful force is placed on the line, SafeBreak intentionally separates to remove the damaging force and protect the patient's IV. When SafeBreak separates, valves on both ends of the device close preventing medicine spills from the pump and blood loss from the patient. The separated device also causes the IV pump to alarm, notifying the nursing staff to check on the patient. To replace SafeBreak, each component is unthreaded from the line, a new sterile SafeBreak is installed, and the patient's infusion is restarted. Patients avoid additional needlesticks, nurses save time, and hospitals save money1. It's a win-win-win.
More on Aussie Journal
- Understanding Commercial Air Conditioning Systems
- Cosca and Hogan & Associates Join Forces to Strengthen Accounting Support for Regional Queensland
- NBA Champion Lamar Odom Launches Anti-Addiction Meme Coin, Ushering in a Disruptive Innovation in Web3
- Aureli Construction Sets the Standard for Seamless Home Additions in Greater Boston
- ScreenPoints Puts Film Investors in the Credits—and in the Money With New FinTech Platform
Ryan Brick, VP of Sales, stated, "This is a large milestone for our company and a steppingstone towards our mission of reducing IV-line failure. With SafeBreak's clearance for all vascular lines, we can help protect every patient user and every IV line throughout the entire country. Sales momentum has continued to build since our pediatric clearance this past May, and now, the expansion of SafeBreak's indications will bring even more interest and help introduce the product to more hospitals. We will continue to work to make SafeBreak the standard of care for every hospital and patient."
Vance Clement, CEO of Lineus Medical, stated, "What an exciting time for SafeBreak Vascular and Lineus Medical! We recently gained licence in Canada and now FDA clearance for all medical lines for patients to two weeks of age and up in the United States. SafeBreak is a simple device that can make a big difference for nurses and hospitals. These new indications are for more complicated and more expensive IV lines that are used for long-term infusions, cancer patients, and in emergency situations. SafeBreak has been shown to work well on peripheral IVs, and now it can be used by nurses to protect these more expensive lines on more vulnerable patient populations. With these additional indications, we can expand the benefit of SafeBreak throughout the entire hospital."
More on Aussie Journal
- Pathways to Adulthood Conference May 17 at Melville Marriott Honoring NYS Assembly Member Jodi Giglio, Suffolk County Legislator Nick Caracappa
- Adster Techologies awarded US Patent for breakthrough innovation in reducing latency in Ad Serving
- This Mother's Day, Give the Gift of Comfort Climate Green Offers Big Discounts on Air Conditioning
- Robert Fabbio Inducted into the Austin Technology Council Hall of Fame
- Cybersecurity is Protecting Your Personal Information and Your Portfolio
ABOUT LINEUS MEDICAL
Lineus Medical is the developer of a break-away technology that is proven to reduce IV restarts and IV complications1. Our mission is to remove the pains associated with medical lines. More information about Lineus Medical can be found on the company website: www.lineusmed.com or you can follow the company on Facebook or LinkedIn.
1. Data on File
MKG-0237 Rev 00
Source: Lineus Medical
0 Comments
Latest on Aussie Journal
- Ditch Micromanagement: New Leadership Book for Results-Driven, Accountability-Based Teams
- Jay Tapp was named Managing Director in British Columbia
- Hubei Heavy Equipment Makes a Striking Appearance at CIMT and Competes with International Brands
- 20 Patents Issued Worldwide, Cementing Company Leadership. First Ever Cable-Free 12-Lead ECG: HeartBeam, Inc. (Stock Symbol: BEAT)
- NASDAQ Uplisting for Higher Market Exposure and Wide Corporate Benefits to AI Boosted Marketing Company On Track Towards $1 Billion Revenue by 2027
- Congressional Men's Health Caucus Shows Bipartisan Consensus and Focus on Prevention, Mental Health, and Closing the Lifespan Gap
- DuoKey, Axiomtek and Blue Edge Network Partner to Enhance Smart Cities with Privacy-Preserving Urban Safeguarding and Fleet Management
- Climate Green: Melbourne's Trusted VEU Accredited Provider Making Electrification Easy with Expert
- Surf Coast Region Emerges as Victoria's Entrepreneurial Powerhouse
- Austin Keen Joins WakeFX RopePal as Official Brand Ambassador
- Bonelli Systems Expands Managed IT Services Nationwide, Leveraging Microsoft Azure Expertise
- $4.3 Million Patent Application Waiver Fee Granted by FDA on New Drug Application Fee for Treatment Addressing Suicidal Depression & PTSD: NRX Pharma
- Whistleblower Claims Dental Patient Deaths Likely Due to Book Ban
- xREnergy up as much as +3,094,634% on first day listed on the XRP Ledger. Ticker : $XRE
- Psychiatry's Legacy of Racism and Coercion Highlighted in Restraint Deaths
- New Book 'Cybersecurity Leadership' Guides SME Leaders to Make Smart, Strategic Security Decisions
- "Stop scrolling and start watching" - Beloved film recommendation site Criticker gets a major makeover
- Green Energy Solar Expands with New Offices in Port St. Lucie, West Palm Beach, and Orlando
- Events by Dubsdread Expands Services to The Venue at Lake Lily
- London Gala Emphasizes Trade, FDI and Ongoing Cooperation