Trending...
- The AI Production Shift: Why Game Development Is Entering Its Most Accelerated Phase
- AI Predicts the Most Likely 2026 FIFA World Cup Winner
- Millions of Pandemic Pets Are Reaching Midlife. One App Is Built to Help Them Thrive
Commercial-Ready Ketamine Production, FDA Alignment, and Expanding AI Neurotherapy Platform Position $NRXP at the Center of a Multi-Billion-Dollar Mental Health Transformation
MIAMI - AussieJournal -- NRx Pharmaceuticals (N A S D A Q: NRXP) is rapidly advancing beyond the traditional biotech model, emerging as a commercially prepared, AI-enabled mental health platform at a time when federal policy, regulatory clarity, and technological innovation are converging in its favor.
The company's trajectory has already been defined by a rare alignment of catalysts—most notably the April 18, 2026 Presidential Executive Order accelerating approval of breakthrough therapies for serious mental illness. This directive is expected to expand the use of real-world evidence, compress regulatory timelines, and introduce priority mechanisms that directly benefit companies like NRx, positioning it within a policy-supported pathway toward faster approvals and broader adoption.
Commercial Inflection Point: Manufacturing Now Underway
Layered on top of this macro tailwind, NRx has taken a decisive operational step forward with the initiation of its first commercial-scale manufacturing order for preservative-free ketamine—positioning the company ahead of anticipated FDA approval under its ANDA pathway expected in Summer 2026.
This marks a clear shift from development to execution.
Key manufacturing advantages include:
NRx's formulation also eliminates Benzethonium Chloride, a preservative no longer permitted in new drugs, creating a modern, differentiated product profile aligned with evolving regulatory standards.
Federal Policy Alignment Creates a Powerful Tailwind
The Presidential directive to accelerate psychedelic and breakthrough therapies is more than a headline—it represents a structural shift in how mental health treatments are approved and deployed.
More on Aussie Journal
NRx is already aligned with this framework, positioning itself to benefit from:
This transforms the company's regulatory pathway into one that is increasingly policy-supported and time-efficient.
FDA Alignment Anchors the Investment Thesis
At the core of the story is a rare regulatory breakthrough.
NRx has secured agreement from the FDA to proceed with its New Drug Application for NRX-100 (preservative-free ketamine) using existing clinical trial data combined with real-world evidence—eliminating the need for additional trials.
This enables a targeted NDA submission as early as June 2026 while expanding the indication to include treatment-resistant depression with suicidality.
Regulatory positioning highlights:
Dual FDA Pathways Create Layered Upside
NRx is uniquely positioned with two concurrent regulatory opportunities.
While the NDA pathway represents long-term value creation, the ANDA pathway—now supported by active manufacturing—introduces near-term commercialization potential.
Dual catalyst structure:
This parallel progression is rare and significantly enhances visibility for investors.
From Biotech to Platform: AI + Neurotechnology Expansion
NRx is no longer a single-product company—it is building an integrated platform combining pharmaceuticals, neurostimulation, and artificial intelligence.
Through its NRx Defense Systems subsidiary, the company is advancing robotic-enabled Transcranial Magnetic Stimulation (TMS) combined with neuroplastic drug therapy, targeting military and first responder populations.
At the same time, AI integration is transforming care delivery.
Platform expansion includes:
Commercial Infrastructure Already Generating Revenue
More on Aussie Journal
NRx is not waiting for approval to build its business.
Through HOPE Therapeutics, the company has established a growing network of interventional psychiatry clinics delivering ketamine therapy, TMS, and neuroplastic treatments.
These clinics are:
This creates a ready-made commercialization engine.
Financial Position Supports Execution
NRx has strengthened its balance sheet through reduced operating expenses, elimination of debt, and sufficient capital to support operations through 2026.
This provides the flexibility to execute across manufacturing, regulatory, and commercialization initiatives simultaneously.
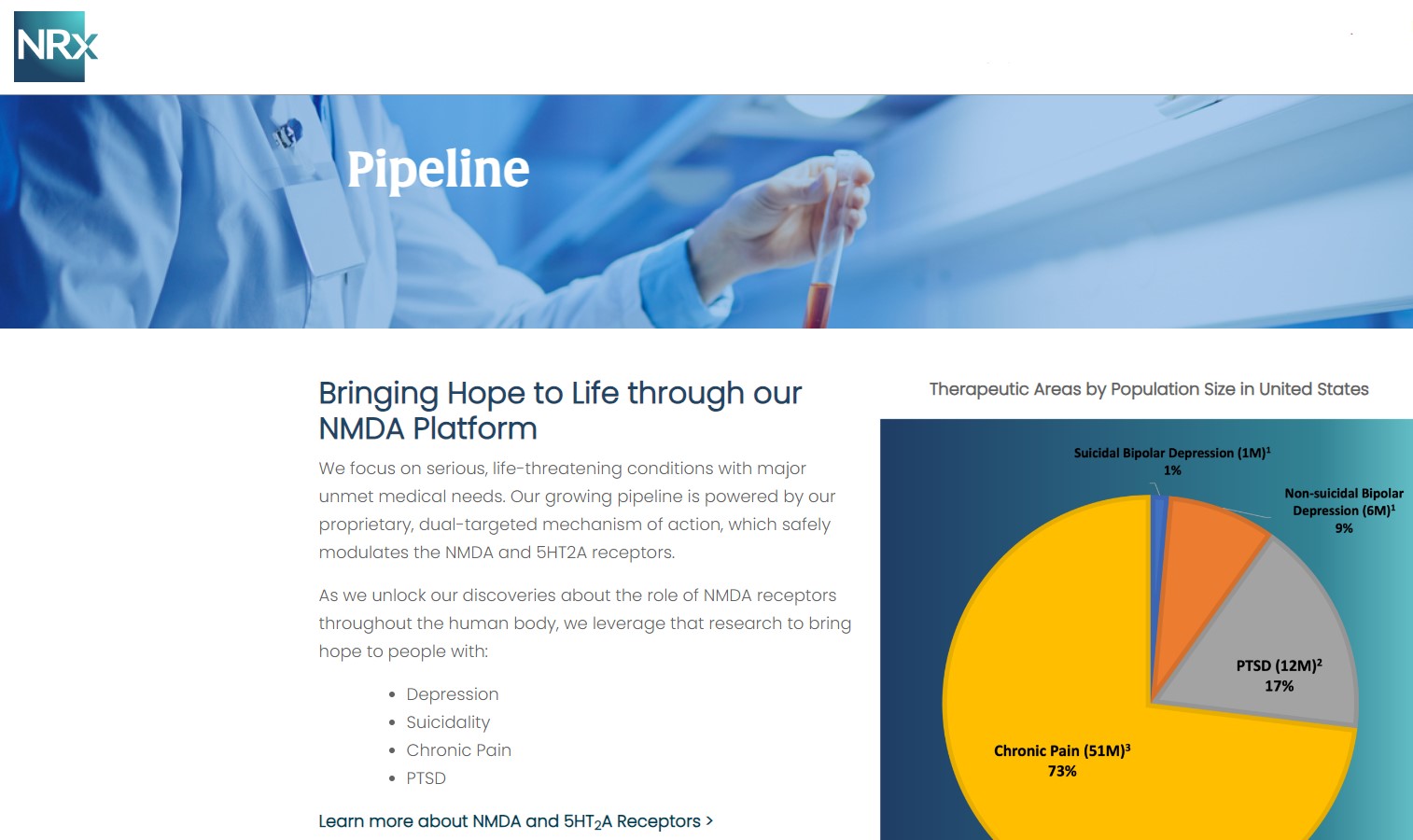
A Massive, Urgent Market Opportunity
NRx is targeting one of the most critical unmet needs in healthcare.
With ketamine shortages persisting and demand for rapid-acting therapies accelerating, $NRXP is entering the market at a time of both urgency and opportunity.
Final Take: From Catalyst Story to Execution Story
NRx Pharmaceuticals is no longer simply approaching catalysts—it is activating them across multiple fronts simultaneously.
With:
$NRXP represents a rare convergence of timing, technology, and execution.
This is no longer a company waiting for approval—
It is a company preparing to deliver at scale.
For more information on $NRXP visit: https://www.nrxpharma.com/ and https://compasslivemedia.com/case-study/nrx-pharmaceuticals/
Company Contact
NRx Pharmaceuticals, Inc. (N A S D A Q: NRXP)
Matthew Duffy – Chief Business Officer
Phone: (484) 254-6134
Email: mduffy@nrxpharma.com
DISCLAIMER: https://corporateads.com/disclaimer/
Disclosure listed on the CorporateAds website
The company's trajectory has already been defined by a rare alignment of catalysts—most notably the April 18, 2026 Presidential Executive Order accelerating approval of breakthrough therapies for serious mental illness. This directive is expected to expand the use of real-world evidence, compress regulatory timelines, and introduce priority mechanisms that directly benefit companies like NRx, positioning it within a policy-supported pathway toward faster approvals and broader adoption.
Commercial Inflection Point: Manufacturing Now Underway
Layered on top of this macro tailwind, NRx has taken a decisive operational step forward with the initiation of its first commercial-scale manufacturing order for preservative-free ketamine—positioning the company ahead of anticipated FDA approval under its ANDA pathway expected in Summer 2026.
This marks a clear shift from development to execution.
Key manufacturing advantages include:
- Completed GMP audits and validated registration batches
- Initial capacity designed to support meaningful first-year revenue
- Rapid scalability via blow-fill-seal process (10x traditional output)
- U.S.-based production addressing ongoing ketamine supply shortages
NRx's formulation also eliminates Benzethonium Chloride, a preservative no longer permitted in new drugs, creating a modern, differentiated product profile aligned with evolving regulatory standards.
Federal Policy Alignment Creates a Powerful Tailwind
The Presidential directive to accelerate psychedelic and breakthrough therapies is more than a headline—it represents a structural shift in how mental health treatments are approved and deployed.
More on Aussie Journal
- Registered NDIS Provider Livara Care Expands Disability Support Services Across Sydney
- CAPHRA warns Southeast Asia not to repeat Australia's nicotine policy failure
- Milo3D.ai Launches Free AI 3D Model Generator That Turns Text and Images Into Game-Ready 3D Assets in Seconds
- UK Financial Ltd Executes 100% Success Rate on All ERC-3643 Transfers to Coin Holders of MayaCat Regulated Security Token and Maya Preferred PRA
- Blank Space: The Unofficial Taylor Swift Tribute Brings Eras Tour Magic To Cities Across America
NRx is already aligned with this framework, positioning itself to benefit from:
- Expanded acceptance of real-world evidence
- Faster FDA review timelines
- Potential access to priority voucher programs
This transforms the company's regulatory pathway into one that is increasingly policy-supported and time-efficient.
FDA Alignment Anchors the Investment Thesis
At the core of the story is a rare regulatory breakthrough.
NRx has secured agreement from the FDA to proceed with its New Drug Application for NRX-100 (preservative-free ketamine) using existing clinical trial data combined with real-world evidence—eliminating the need for additional trials.
This enables a targeted NDA submission as early as June 2026 while expanding the indication to include treatment-resistant depression with suicidality.
Regulatory positioning highlights:
- No additional trials required
- Fast Track designation already granted
- 70,000+ patient real-world dataset supporting efficacy
- Expanded label strategy increasing addressable market
Dual FDA Pathways Create Layered Upside
NRx is uniquely positioned with two concurrent regulatory opportunities.
While the NDA pathway represents long-term value creation, the ANDA pathway—now supported by active manufacturing—introduces near-term commercialization potential.
Dual catalyst structure:
- ANDA approval (anticipated Summer 2026) → near-term revenue
- NDA submission (target June 2026) → transformational upside
This parallel progression is rare and significantly enhances visibility for investors.
From Biotech to Platform: AI + Neurotechnology Expansion
NRx is no longer a single-product company—it is building an integrated platform combining pharmaceuticals, neurostimulation, and artificial intelligence.
Through its NRx Defense Systems subsidiary, the company is advancing robotic-enabled Transcranial Magnetic Stimulation (TMS) combined with neuroplastic drug therapy, targeting military and first responder populations.
At the same time, AI integration is transforming care delivery.
Platform expansion includes:
- AI-driven patient monitoring ("Depression Thermometer")
- Real-time treatment optimization
- Robotic, precision-guided neurostimulation systems
- Potential non-dilutive government and defense funding channels
Commercial Infrastructure Already Generating Revenue
More on Aussie Journal
- Love Must Be the Guide: Live Good Shares a Message of Humanity, Compassion and Hope
- D.R. Crotzer Announces A New Science Fiction Book Series Exploring Life Energy, Dreams, and the Mystery of Existence
- Color Card Administrator Highlights Growing Enterprise Demand for Operational Infrastructure in Business Card Identity Governance
- American Properties Celebrates Grand Opening and Ribbon Cutting Ceremony at Heritage at South
- Crosswalk Ministries USA Announces 2026 Child and Family Well-Being Conference in Stockbridge, Georgia
NRx is not waiting for approval to build its business.
Through HOPE Therapeutics, the company has established a growing network of interventional psychiatry clinics delivering ketamine therapy, TMS, and neuroplastic treatments.
These clinics are:
- Operational and revenue-generating
- Supported by government programs, insurers, and private pay
- Positioned for rapid scale post-approval
This creates a ready-made commercialization engine.
Financial Position Supports Execution
NRx has strengthened its balance sheet through reduced operating expenses, elimination of debt, and sufficient capital to support operations through 2026.
This provides the flexibility to execute across manufacturing, regulatory, and commercialization initiatives simultaneously.
A Massive, Urgent Market Opportunity
NRx is targeting one of the most critical unmet needs in healthcare.
- Over 13 million Americans seriously consider suicide annually
- Rising global rates of treatment-resistant depression and PTSD
With ketamine shortages persisting and demand for rapid-acting therapies accelerating, $NRXP is entering the market at a time of both urgency and opportunity.
Final Take: From Catalyst Story to Execution Story
NRx Pharmaceuticals is no longer simply approaching catalysts—it is activating them across multiple fronts simultaneously.
With:
- Commercial manufacturing now underway
- Federal policy accelerating approvals
- FDA-aligned pathways requiring no new trials
- Dual regulatory opportunities
- Expanding AI and neurotechnology platform
- Revenue-generating clinical infrastructure
$NRXP represents a rare convergence of timing, technology, and execution.
This is no longer a company waiting for approval—
It is a company preparing to deliver at scale.
For more information on $NRXP visit: https://www.nrxpharma.com/ and https://compasslivemedia.com/case-study/nrx-pharmaceuticals/
Company Contact
NRx Pharmaceuticals, Inc. (N A S D A Q: NRXP)
Matthew Duffy – Chief Business Officer
Phone: (484) 254-6134
Email: mduffy@nrxpharma.com
DISCLAIMER: https://corporateads.com/disclaimer/
Disclosure listed on the CorporateAds website
Source: CorporateAds
0 Comments
Latest on Aussie Journal
- XRPPower Continues Strengthening Its Global AI-Powered Blockchain Ecosystem
- Lick Introduces Pineapple Flavored Massage Oil — A Tropical Date Night Favorite Available on Amazon
- FutureLot Powers ADU Wizard for Massachusetts Clean Energy Center's Statewide ADU Resource Center
- ICT Innovations Releases ICTPBX Community Edition as Open Source Under Mozilla Public License 2.0
- Maryland Personal Injury Firm Earns National Recognition in 2026 ELA Awards
- Robert J. Bradshaw's AYE is a Gripping Dual Reality Thriller Exploring the Increasingly Blurred Line Between Humanity and Technology
- Bangxing Silicone Revolutionizes Silicone Baby Product Partnerships: Low MOQ Support + VIP Long-Term Win-Win Programs
- SteelTree Announces Launch of Its Operational Decision Intelligence Service
- Advanced AI Capabilities Reflected by Upcoming Company Name and Stock Symbol Change for Evolving Pre-Owned Boat Dealer: Off The Hook YS: N Y S E: OTH
- AI-Driven Defense Expansion, Autonomous Systems and Israeli Aerospace Manufacturing Platform: VisionWave Holdings (N A S D A Q: VWAV)
- AI Predicts the Most Likely 2026 FIFA World Cup Winner
- Web App Development Services by Omnific IT – Your Gateway to Digital Solutions
- The AI Production Shift: Why Game Development Is Entering Its Most Accelerated Phase
- Money for nothing: Aussies score cashback on household bills
- World-First AI Humanoid Robot Debuts on Cherie Barber's Ground-breaking Australian Reno Show
- Millions of Pandemic Pets Are Reaching Midlife. One App Is Built to Help Them Thrive
- New Survey Reveals America's Most Feared Bridges for Cyclists — Golden Gate Tops the List
- Raymond Lavine, Extended Care Benefits Advisor and Author, to Appear on National Television Series Moving America Forward
- New 24-Hour Emergency Plumbing Service Launches in Bondi Beach
- NaturismRE Launches Structured Nudism & Naturism Encyclopedia, Aiming to Reframe Public Understanding