Trending...
- EOFY OFFER - Register 4 Delegates, 5th Delegate FREE 2026 INDIGENOUS CONFERENCES - 121
- Lineus Medical Completes Financial Restructuring with KMF Investments- Launching a New Era for SafeBreak
- Zeus and the Thunderers of the Ancient World
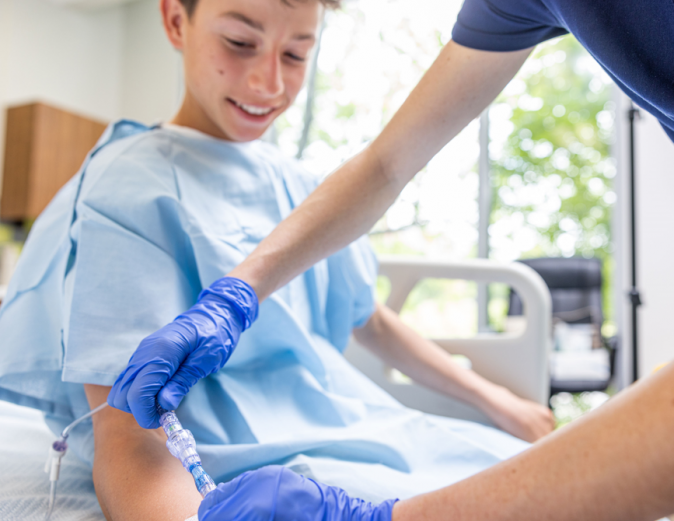
FAYETTEVILLE, Ark. - AussieJournal -- Lineus Medical, a medical device company focused on reducing IV complications, announced that the company's flagship product, SafeBreak Vascular, has received clearance from the U.S. Food and Drug Administration (FDA) for use in pediatric patients. SafeBreak Vascular has been shown in adult clinical studies to reduce IV complications with its innovative breakaway technology1.
SafeBreak Vascular is the first breakaway device cleared by the FDA and proven to reduce IV complications in adults by 44%1. It fits between the IV tubing from the IV pump and the patient's IV access point at the arm. When there is a damaging force on the line, SafeBreak separates to protect the patient's IV. This innovative feature helps prevent accidental dislodgement of the catheter, as well as other IV complications caused by damaging forces on the IV line1. This is especially important for pediatric patients who may be more active, less inclined to follow instructions, and less aware of their surroundings.
More on Aussie Journal
"We are incredibly proud to receive clearance from the FDA for the use of SafeBreak Vascular with pediatric patients," said Vance Clement, CEO of Lineus Medical. "SafeBreak provides a simple, effective solution for preventing IV complications, which reduces IV restarts and additional needlesticks for patients. Now, it doesn't matter if the patient is 1 or 100 years old, SafeBreak is cleared for use on all peripheral IVs. Our goal is to have a SafeBreak on every IV line, everywhere in the world, and this additional clearance lets us help one of the most vulnerable populations that needs it the most, our kids."
ABOUT LINEUS MEDICAL
Lineus Medical is the developer of a unique, patented, break-away technology that is designed to work with all types of medical tubing. Complications with feeding tubes, IV lines, urinary catheters and any other type of medical tubing may be reduced as breakaway devices separate to protect the patient. More information about Lineus Medical can be found on the company website: www.lineusmed.com or you can follow the company on Twitter or LinkedIn.
1 Dipper Study Data on File
MKG-0194 REV 00
SafeBreak Vascular is the first breakaway device cleared by the FDA and proven to reduce IV complications in adults by 44%1. It fits between the IV tubing from the IV pump and the patient's IV access point at the arm. When there is a damaging force on the line, SafeBreak separates to protect the patient's IV. This innovative feature helps prevent accidental dislodgement of the catheter, as well as other IV complications caused by damaging forces on the IV line1. This is especially important for pediatric patients who may be more active, less inclined to follow instructions, and less aware of their surroundings.
More on Aussie Journal
- Greenland Mines Ltd (N A S D A Q: GRML) Advances Strategic Growth Initiatives as Critical Minerals Demand Accelerates
- Entering the $69 Billion Animal Health Market, Delivering Record Growth, AI-Driven Healthcare Innovation, and Targeting $200 Million Revenue by 2029
- $97.9 Million Q1 Revenue Growth Reinforces Transformation Into a Global AI & Digital Services Powerhouse: IQSTEL, Inc. (N A S D A Q: IQST)
- Brisbane Consultancy Launches AI Search Visibility Platform for Local Businesses
- Boston Industrial Solutions Launches Natron® 348 UV Inkjet Ink for Epson S3200 Print Heads
"We are incredibly proud to receive clearance from the FDA for the use of SafeBreak Vascular with pediatric patients," said Vance Clement, CEO of Lineus Medical. "SafeBreak provides a simple, effective solution for preventing IV complications, which reduces IV restarts and additional needlesticks for patients. Now, it doesn't matter if the patient is 1 or 100 years old, SafeBreak is cleared for use on all peripheral IVs. Our goal is to have a SafeBreak on every IV line, everywhere in the world, and this additional clearance lets us help one of the most vulnerable populations that needs it the most, our kids."
ABOUT LINEUS MEDICAL
Lineus Medical is the developer of a unique, patented, break-away technology that is designed to work with all types of medical tubing. Complications with feeding tubes, IV lines, urinary catheters and any other type of medical tubing may be reduced as breakaway devices separate to protect the patient. More information about Lineus Medical can be found on the company website: www.lineusmed.com or you can follow the company on Twitter or LinkedIn.
1 Dipper Study Data on File
MKG-0194 REV 00
Source: Lineus Medical
Filed Under: Technology
0 Comments
Latest on Aussie Journal
- P-Wave Classics Opens Pre-Orders for Volume II of Robert Bage's Hermsprong
- 3 Ways to Style a Plain Bangle Bracelet
- Tuckwell Machinery Launches New Range of Woodworking Machinery
- A Brave Little Hero with Four Paws
- Revenue Optics Expands Its Private Equity Practice as Sponsors Move Inside Sales to the Center of Distribution Value Creation
- Ecuador Freedom Launches First Scheduled Motorcycle Tour of Northern Peru's Lost Kingdoms
- Lineus Medical Completes Financial Restructuring with KMF Investments- Launching a New Era for SafeBreak
- Neuro Recovery Institute Showcases Emerging Immersive Neuro-Rehabilitation Technology at Clinical Innovation Open House
- Community, Conservation & Waterwise Inspiration Bloom on June 6
- Industrial and systems engineers celebrate key leaders in the field at IISE Annual Conference
- Cosanostra Miami Rises as the Best Latin Nightclub in Miami in Under Two Years From its Opening
- CCHR Leader's 50-Year Fight for Psychiatric Drug Victims Gains National Momentum
- humAIn launches as indie media and events platform to help Australian marketing industry navigate AI
- Author Releases 7-Day Screen Time Reset for Families as Teachers Worldwide Report Children "Struggling to Grasp Basic Concepts"
- Men's Health Month Begins with Record Proclamations, AP News Coverage, & National Momentum for Men's Health
- Research & Data Reveals Back Pain's Mental, Physical and Financial Toll
- AdvisorVault Adds Social Media Archiving to its Consolidated D3P Service
- UK Financial Ltd Audits Full Ethereum Architecture Verifies Corporate Wallets and 19-Token Ecosystem Ahead of CoinMarketCap Filing for Global Ranking
- Creative Investment Research Analysis Finds Slower GDP Growth, Rising Inflation
- TechHouse Earns Highly Selective Microsoft Support Badge